This lecture can be viewed on YouTube site: Life Scinence Lectures for you
https://youtu.be/CV7Jy_q__ts
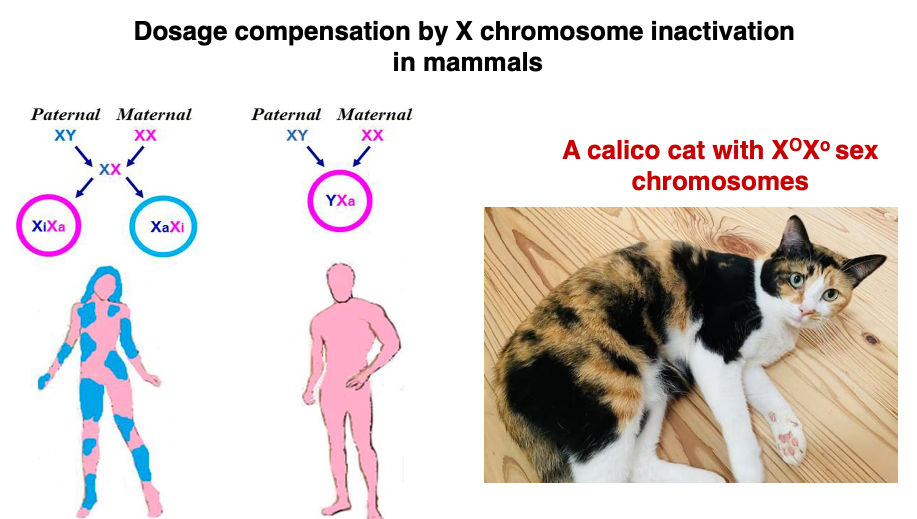
1. This lecture explains gene dosage compensation through X chromosome inactivation in mammals.
- Abnormalities in sex chromosome composition and associated symptoms.
2. Autosomal trisomy and its symptoms
3. Random inactivation of X chromosomes.
4. Various mechanisms of X chromosome dosage compensation.
5. Discovery of the X Inactivation Center (XIC).
6. Xist and Tsix non-coding long RNAs
7. Unequal distribution of CTCF through pairing and separation of XIC.
8. Mechanism determining which X chromosome will be inactivated.
Keywords: pseudoautosomal region, PAR, XXY, XXX, XO, autosomal trisomy, Mary Lyon’s hypothesis, calico cat coat color, X inactivation center (XIC), Tsix RNA, Xist RNA, epigenetic histone modifications
2. Chromosomal composition of human somatic cells
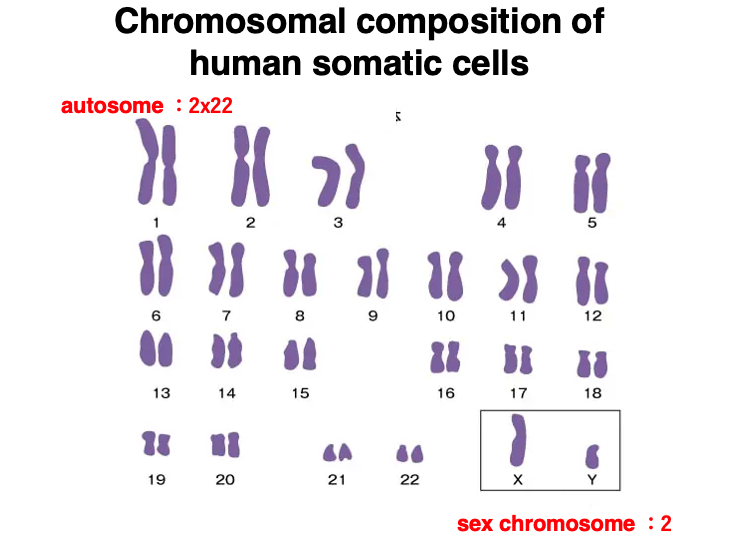
This figure shows the chromosomal composition of human somatic cells.
Human somatic cells are 2n (diploid), so there are pairs of autosomes numbered from 1 to 22 in order of decreasing size.
As for sex chromosomes, females have two X chromosomes, while males have one X chromosome and one Y chromosome.
3. Comparison of X chromosome and Y chromosome
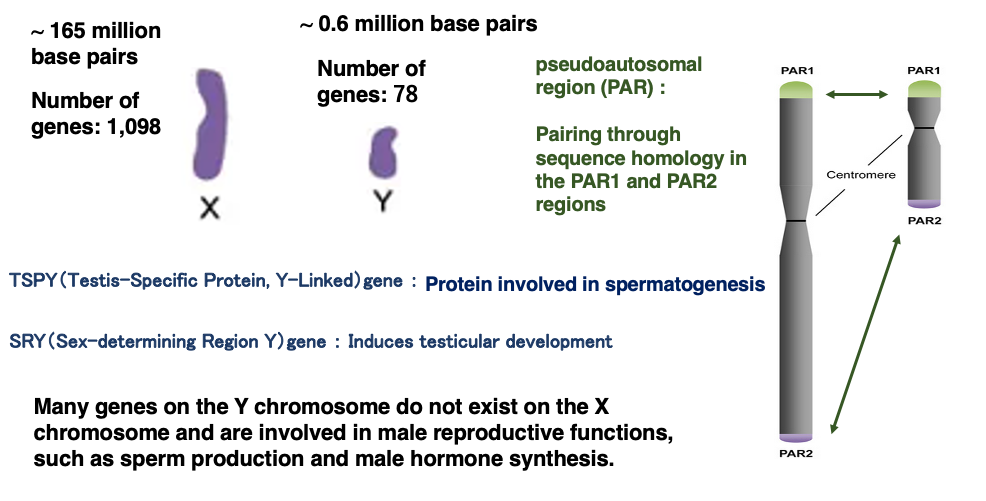
While the X chromosome is composed of approximately 165 million base pairs, the Y chromosome consists of about 60 million base pairs. Consequently, the size of the Y chromosome is less than one-third of the X chromosome.
The X chromosome encodes 1,098 genes, while the Y chromosome encodes 78 genes.
The Y chromosome contains genes related to sperm formation (SRY gene) and male hormone production (TSPY gene). These genes are not present on the X chromosome.
Although the X and Y chromosomes differ greatly in size and internal base sequence, they pair up during meiosis like autosomal chromosome pairs. The pairing of X and Y chromosomes is possible due to the presence of pseudoautosomal regions (PAR1 and PAR2) at the ends of the X and Y sex chromosomes. These regions contain highly conserved sequences shared by both chromosomes.
4. Abnormalities in sex chromosome composition and their effects

When a fertilized egg has an abnormal number of X chromosomes, mild abnormalities may occur, but no abnormalities that significantly interfere with daily life appear. Women with only one X chromosome exist at a rate of 1 in 1,000 to 2,000 people. In this case, there are no major problems in normal life, but there is a tendency for short stature.
Women with three X chromosomes also exist at a rate of about 1 in 1,000 people. In this case as well, there are almost no physical characteristics.
Men with an XXY sex chromosome composition also exist at a rate of about 1 in 1,000 people. In this case too, there are no problems in daily life.
On the other hand, fertilized eggs with only Y or YY sex chromosome compositions result in spontaneous miscarriage very early in development, so such humans do not exist. This is because the X chromosome contains many genes necessary not only for sex determination but also for life maintenance, and chromosomes with only autosomes and Y chromosomes do not have enough genetic information to form an individual.
Conversely, there are no genes necessary for life maintenance on the Y chromosome. Therefore, individuals with XO or XX sex chromosomes can survive. From these facts, it is clear that autosomes and X chromosomes contain all the genes necessary to form a female, and genes on the Y chromosome are needed to form an individual with male characteristics based on a female.
It is understood that males are created by modifying females. In somatic cells, one X chromosome is in a state where genes can be transcribed, while the other X chromosome is highly condensed and inactivated, which is the normal state. For this reason, XO individuals do not have significant differences from XX individuals. Also, in XXX individuals, there is a mechanism by which two of the three X chromosomes are inactivated, so no major abnormalities are observed.
5. Effects of autosomal Trisomy
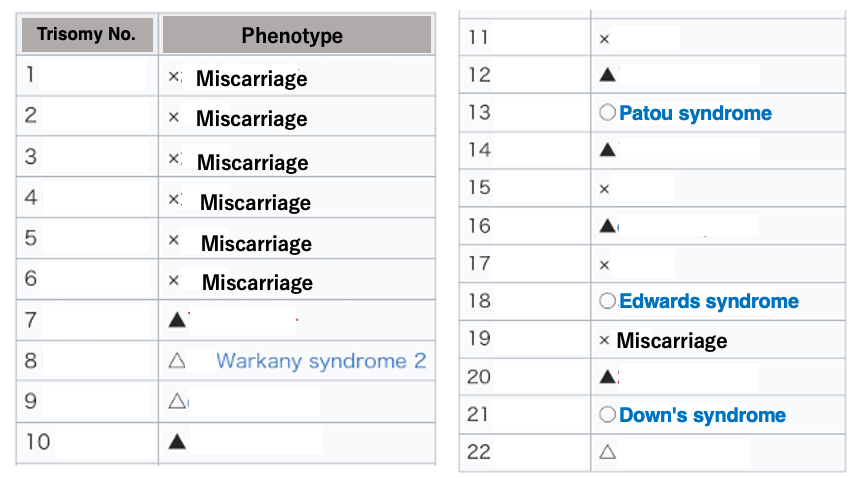
Next, I will explain the effects of having three copies of specific autosomes (trisomy). Autosomes are numbered in order of decreasing size. When large autosomes, such as chromosomes 1 to 6, become trisomy, it is known to result in miscarriage.
On the other hand, when smaller chromosomes become trisomy, various syndromes develop, but miscarriage does not occur. This is thought to be because smaller chromosomes encode a limited number of genes.
In the case of trisomy, there is no deficiency in the genetic information necessary for forming an individual. Nevertheless, trisomy causes various abnormalities because the increased metabolites from specific genes in the cell can have harmful effects on the cell. Unlike the X chromosome, autosomes do not have a mechanism to detect the number of chromosomes and inactivate excess ones.
6. X Chromosome Inactivation
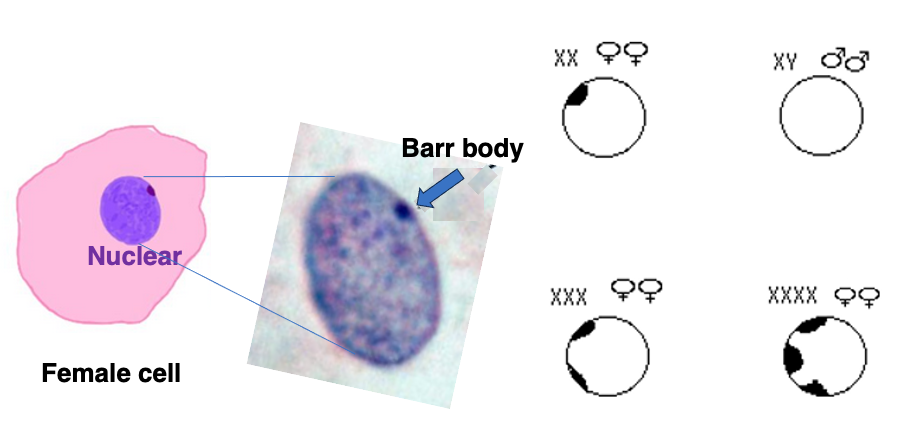
Around 1949, Dr. Barr discovered that when staining chromosomes in female somatic cells, a consistently stained dot could be observed at the cell periphery, which he named the Barr body. Barr bodies are not observed in male somatic cells.
It has been found that the Barr body is one of the two X chromosomes in a condensed state. In the case of XXX cells, two Barr bodies are observed, and in XXXX cells, three Barr bodies are observed.
The Barr body test was one of the sex verification methods used for Olympic athletes from the 1960s to the 1990s.
7. Mary Lyon’s hypothesis

In 1961, Mary Lyon proposed a hypothesis regarding the Barr body, as illustrated in the diagram. This hypothesis was later confirmed to be correct through molecular biological research.
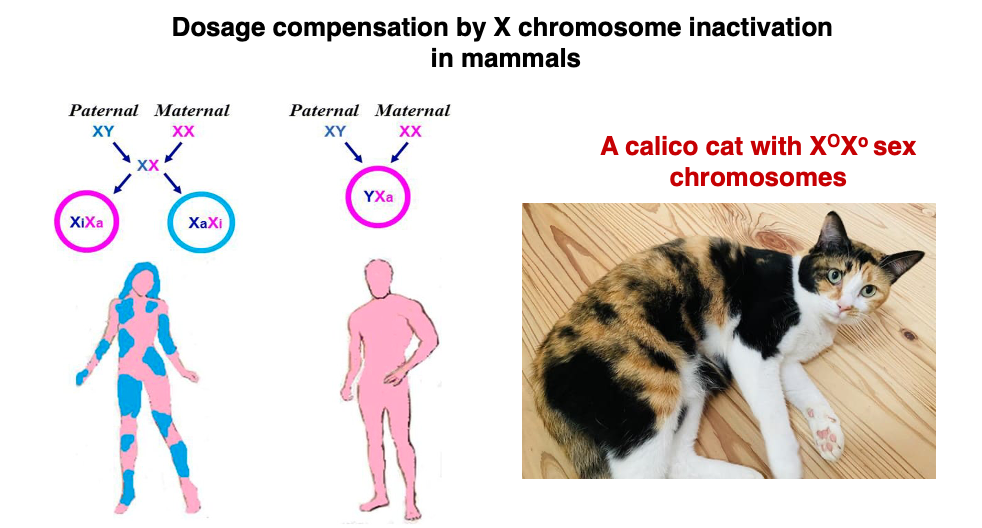
8. Random Inactivation of X Chromosome
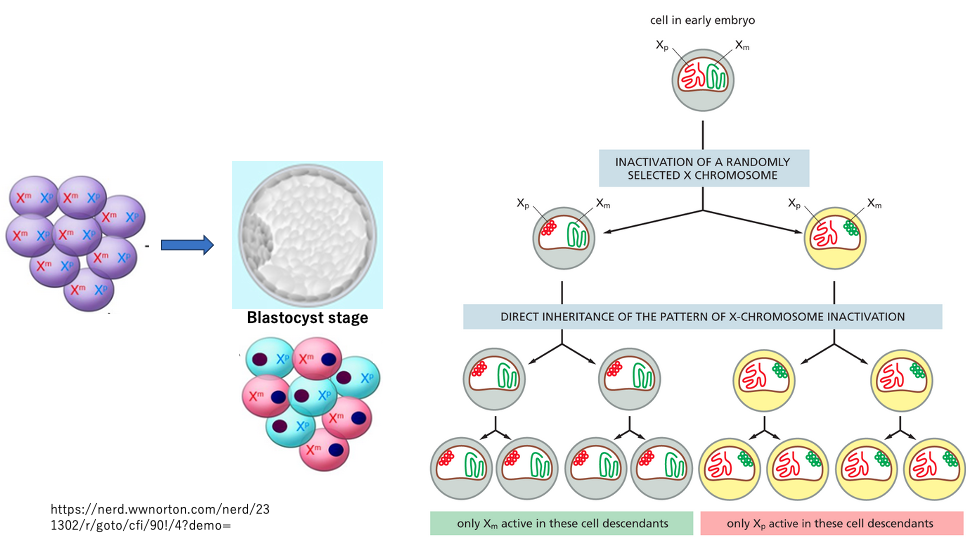
In XX fertilized eggs, the inactivation of either the maternally derived X chromosome or the paternally derived X chromosome occurs randomly.
The determination of which X chromosome will be inactivated takes place during the blastocyst stage, approximately 5 days after fertilization. Once an X chromosome is inactivated, this inactivation (condensation) is maintained through subsequent somatic cell divisions.
As an exception, in the germ cells of the fetus, the condensation of the previously inactivated X chromosome is reversed. Consequently, in the next generation, both sperm and eggs contain activated X chromosomes.
9. X-chromosome Chimera in Adult individuals
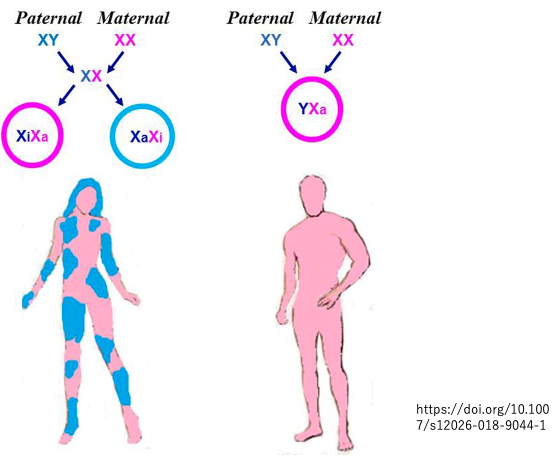
As a result of random X-chromosome inactivation selected during the blastocyst stage being maintained throughout subsequent development, adult female bodies exhibit a patchwork distribution regarding chromosome selection.
10. Gene Expression related to calico Cat coat color

In cats, genes determining fur color are located on the X chromosome. If the dominant gene O is present, black pigment is deposited, while in the case of the recessive gene o, brown pigment is deposited. On the other hand, in regions where the autosomal gene S is expressed, pigment deposition is inhibited, resulting in white fur regardless of the genes on the X chromosome.
Therefore, in the case where a calico cat’s sex chromosomes are XOXo, the black fur regions are where XO is active and the S gene is not expressed. The brown fur regions are where Xo is active and the S gene is not expressed. In the white regions, it is unclear which X chromosome is active, but these are areas where the S gene is expressed.
11. Mechanisms of X chromosome cosage compensation in various organisms
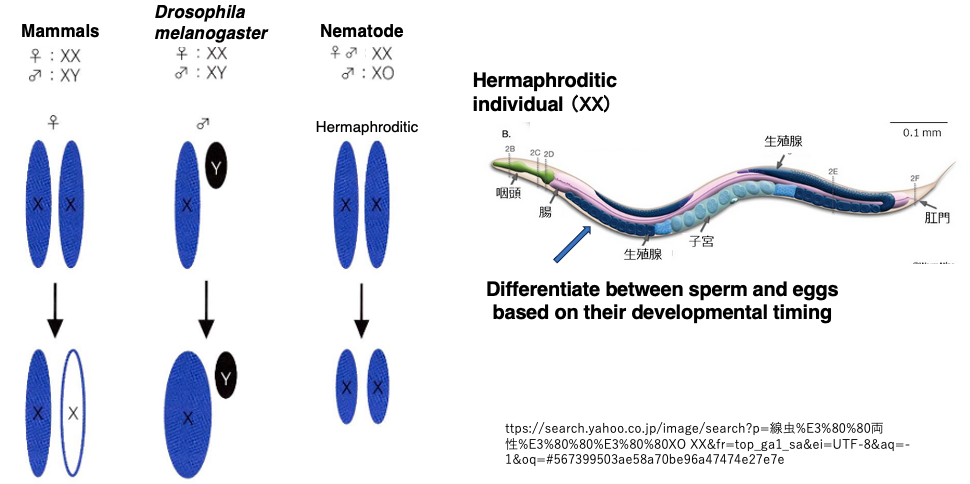
In species with XY sex chromosomes, the mechanisms of sex chromosome dosage compensation vary among different organisms.
In mammals, one of the X chromosomes is completely inactivated, ensuring that the expression level of X in cells with XY is equal to the transcription level from X in cells with XX.
In contrast, in Drosophila (fruit flies), the expression level of X is doubled only in XY males, compensating to match the expression level in XX female cells.
In nematodes (such as C. elegans), XX individuals produce both sperm and eggs (hermaphrodites), while XO individuals are males. In cells with XX, the transcription level of both X chromosomes is halved to match the transcription level in XO cells.
12. Genes expressed on the inactivated X chromosome
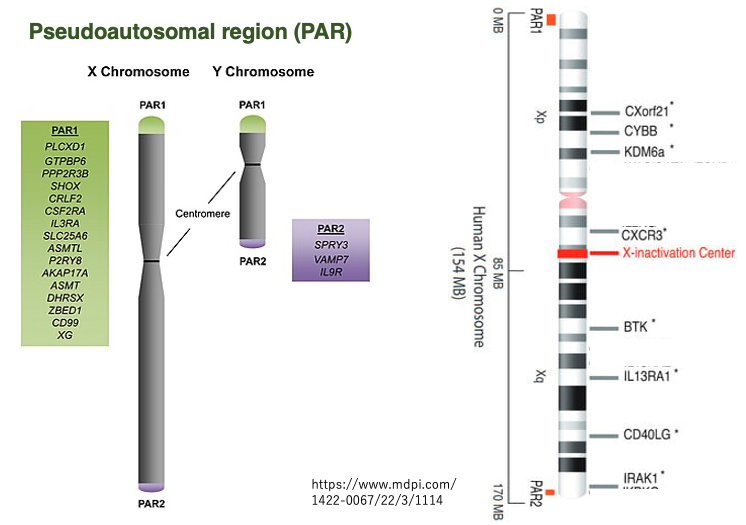
The pseudoautosomal regions,PAR1 and PAR2, are located at both ends of the X and Y sex chromosomes, and these regions are used for X-Y chromosome pairing during meiosis, as previously introduced. Genes in these regions escape inactivation even on the inactivated X chromosome, and are not subject to expression suppression.
The PAR regions are conserved on both X and Y chromosomes and encode common genes, so whether XX or XY, genes in these regions are expressed from two copies, similar to autosomal genes.
In humans, it has been found that a considerable number of genes outside the PAR regions on the X chromosome also escape inactivation and are expressed (about 30% of all X chromosome genes). For such genes, two copies are expressed in XX individuals while only one copy is expressed in XY individuals.
This is the reason for the mild abnormalities observed in individuals with XO (Turner syndrome) or XXY (Klinefelter syndrome) chromosomal compositions.
13. Discovery of the X-inactivation center (XIC)
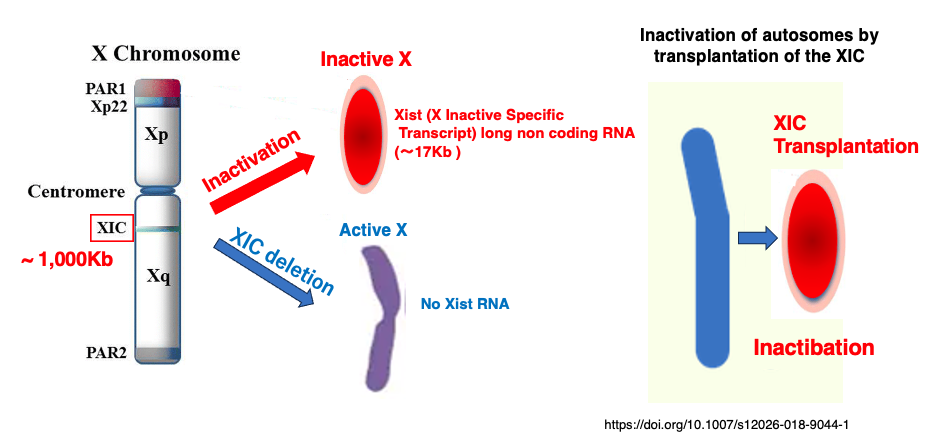
It was found that when a specific region on the X chromosome is deleted, such
a X chromosome does not undergo inactivation. This region was named the X-inactivation center (XIC) region.
This region consists of approximately 1,000 kb. Furthermore, it was discovered that when this region is transplanted to an autosome, that autosome becomes inactivated.
Additionally, a ~17 kb long non-coding RNA was detected, expressed only from the XIC region on the inactive X chromosome. The gene for this RNA was named X Inactive Specific Transcript (Xist). The Xist gene was found to have little to no expression on the active X chromosome.
14. X chromosome Inactivation by Xist RNA attachment
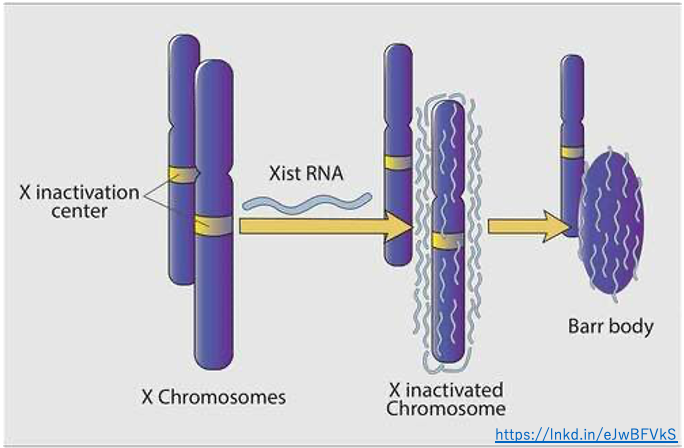
Subsequent studies have shown that the non-coding long RNA, which is a transcript of Xist, attaches to almost the entire X chromosome from which it is transcribed (except for regions such as pseudoautosomal regions).
15. Two non-coding long RNAs encoded within the XIC: Xist and Tsix
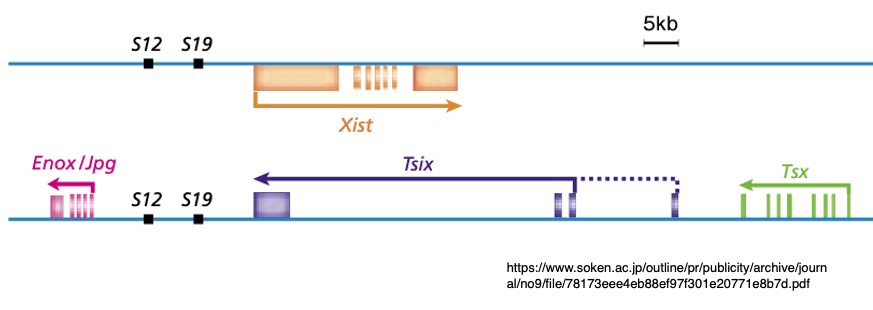
Subsequent research has revealed that in addition to the Xist non-coding long RNA gene, the XIC region contains a gene that transcribes Tsix non-coding long RNA as an antisense transcript from its complementary strand.
Moreover, it is discovered that Tsix RNA functions to suppress the expression of Xist RNA.
16. Determination of the inactive X chromosome through XIC region pairing
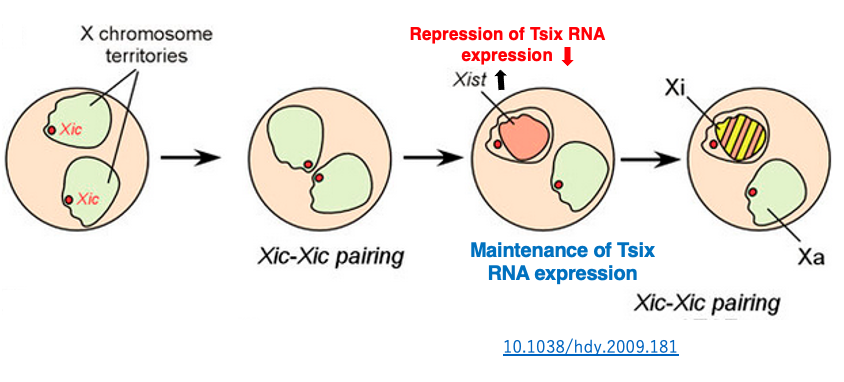
In undifferentiated somatic cells, both Xist and Tsix are expressed at low levels on both X chromosomes. Eventually, a phenomenon of X chromosome pairing is observed. During this time, the XIC region is also observed to pair for about 40 minutes.
It has been discovered that through this transient pairing of the XIC region, the inactivation of one X chromosome is randomly determined. Immediately after pairing, Tsix RNA expression is transiently suppressed on one of the X chromosomes. Due to the decrease in this inhibitory RNA, Xist RNA expression is activated on this X chromosome.
The transcribed Xist RNA covers the surface of that X chromosome, inducing the X chromosome inactivation. On the other hand, on the X chromosome where Tsix RNA expression levels did not change, Xist RNA expression remains at a low level, so inactivation does not occur.
17. Determination of the inactive X chromosome through unequal distribution of XIC-associated proteins
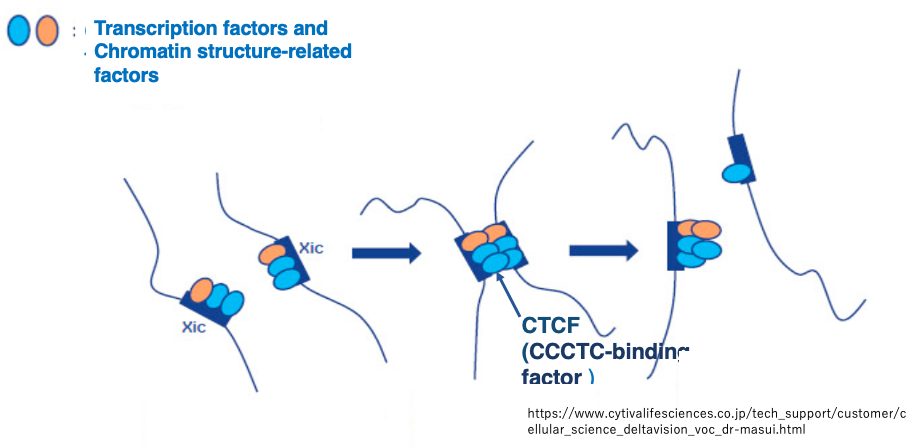
After transient pairing, the XICs separate. It is thought that this pairing and separation results in an unequal distribution of proteins and transcription factors involved in chromatin structure regulation that were attached to the XIC region.
This determines which X chromosome will be inactivated. Among the proteins involved in chromatin structure regulation, CTCF (CCCTC-binding factor) has garnered particular attention.
18. X chromosome Inactivation involving Histone modifications and DNA methylation
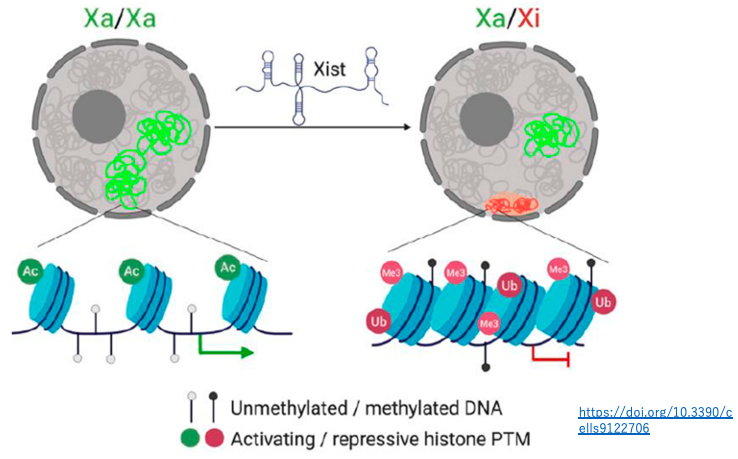
On the inactivated X chromosome, in addition to being covered by Xist RNA, there are several characteristics:
- A higher frequency of repressive histone modifications (H3K27me3, H2AK119ub)
- A lower frequency of histone acetylation
- A higher rate of DNA methylation
Through these epigenetic chromosome modifications, the condensation of the inactivated X chromosome is stably maintained.
19. Process how the Inactive X chromosome is determined
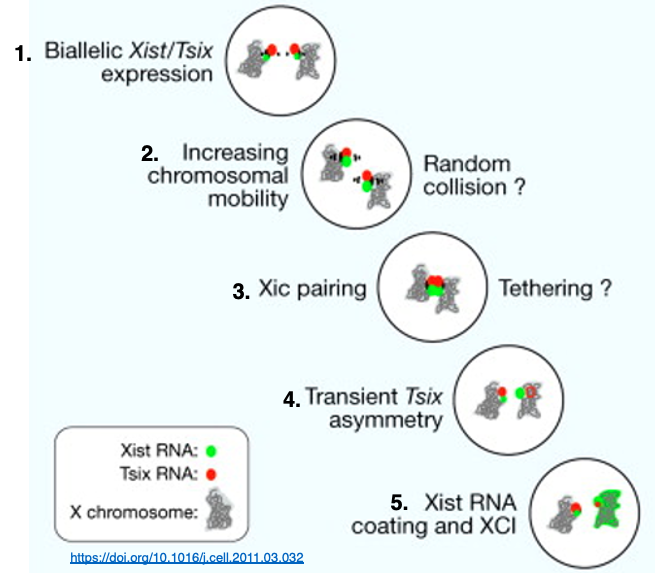
1. In undifferentiated cells, both X chromosomes have low-level expression of Xist and Tsix genes in the XIC (X-inactivation center) region. The X chromosomes are not condensed at this stage.
2. The mobility (fluidity) of X chromosomes within the cell becomes more active, promoting collisions between the X chromosomes.
3. A brief pairing occurs between the XIC regions of the two X chromosomes.
4. The XIC regions separate, leading to an uneven distribution of CTCF and transcription factors. As a result, Tsix RNA expression is suppressed on one X chromosome, which in turn activates Xist RNA expression, leading to the transcription of large amounts of Xist RNA.
5. The X chromosome expressing large amounts of Xist RNA becomes covered entirely by Xist RNA on its surface, initiating high-level chromosome condensation. However, in pseudoautosomal regions, Xist RNA does not attach, allowing these areas to remain in a transcriptionally active state.
コメント